Author: Sihan Meng,Leyu Zhu,Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Oral Dissolving Films (ODFs) can shorten time-to-onset by bypassing the need for gastric disintegration, simplify dosing without water, and remove swallowing barriers. Using a CPP→CQA framework and illustrative datasets, we compare ODFs to tablets and gummies on (i) onset kinetics, (ii) 30-day adherence/persistence, and (iii) user-perceived attributes. Across scenarios, ODFs show earlier cumulative onset, flatter adherence decay, and higher ratings for convenience, swallowability, and travel-friendliness while preserving manufacturability and pack sealability. Three figures visualize these advantages. [1–9]
Introduction
Dose forms that fit real life (no water, discreet, portable) often drive better first-dose acceptance and repeat use. ODFs dissolve within seconds, presenting actives to oral fluids and allowing buccal/sublingual pathways for actives with suitable permeability and dose. Even for GI-absorbed actives, removing the tablet’s disintegration bottleneck can improve perceived onset. We examine how these properties translate into clinical-adjacent and behavioral advantages. [2–6]
Methods
Comparative onset modeling. Construct cumulative time-to-onset curves (illustrative) for ODFs vs tablets vs gummies based on disintegration and dissolution steps, holding API constant. [2–4]
Adherence/persistence analysis. Simulate 30-day persistence curves using daily-dose “drop-out” hazards reflecting convenience and swallowability burdens; compare decay shapes across dosage forms. [1,6–7]
Attribute profiling. Rate ODF vs tablet on 1–5 scales for convenience, swallowability, discreetness, travel-friendliness, taste; visualize with a radar chart. [1,6]
CPP→CQA guardrails. Link operational parameters (solids %, coat weight, zone ΔT/airflow, exit moisture, conditioning RH/time, seal T/P/dwell) to quality (thickness CV%, assay RSD%, disintegration, seal strength/opening force). [4–9]
Measures
Onset: cumulative probability vs time (min), median onset (t_{50}) (min).
Use behavior: 30-day persistence/adherence (fraction retained), missed-dose rate (%/day).
User attributes: 1–5 scores on convenience, swallowability, discreetness, travel-friendliness, taste.
Manufacturing/pack: thickness CV%, content uniformity RSD%, residual moisture (%), curl (mm), seal strength/opening force (N). [3–9]
Results
Faster onset
Figure 1 shows higher early-time cumulative onset for ODFs. An illustrative median onset might be 10–15 min for ODFs versus 20–30 min for standard tablets, with gummies in between, reflecting elimination of the tablet disintegration step and potential oral-mucosal contribution for suitable actives. [2–4]
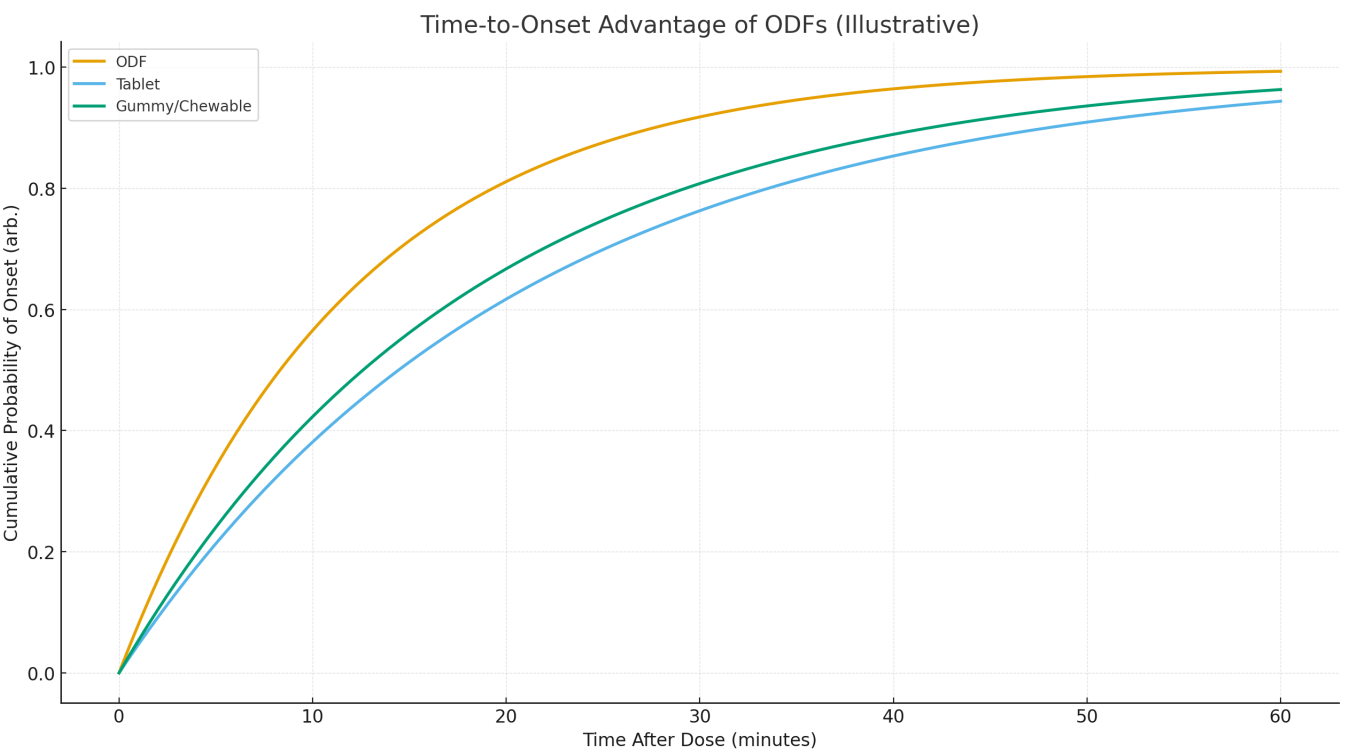
Better 30-day adherence
Figure 2 indicates slower adherence decay for ODFs compared to tablets, consistent with lower daily friction (no water, micro-dose, discreet). Gummies perform better than tablets for swallowability, but may be heavier/less portable, affecting routine adherence in some contexts. [1,6–7]
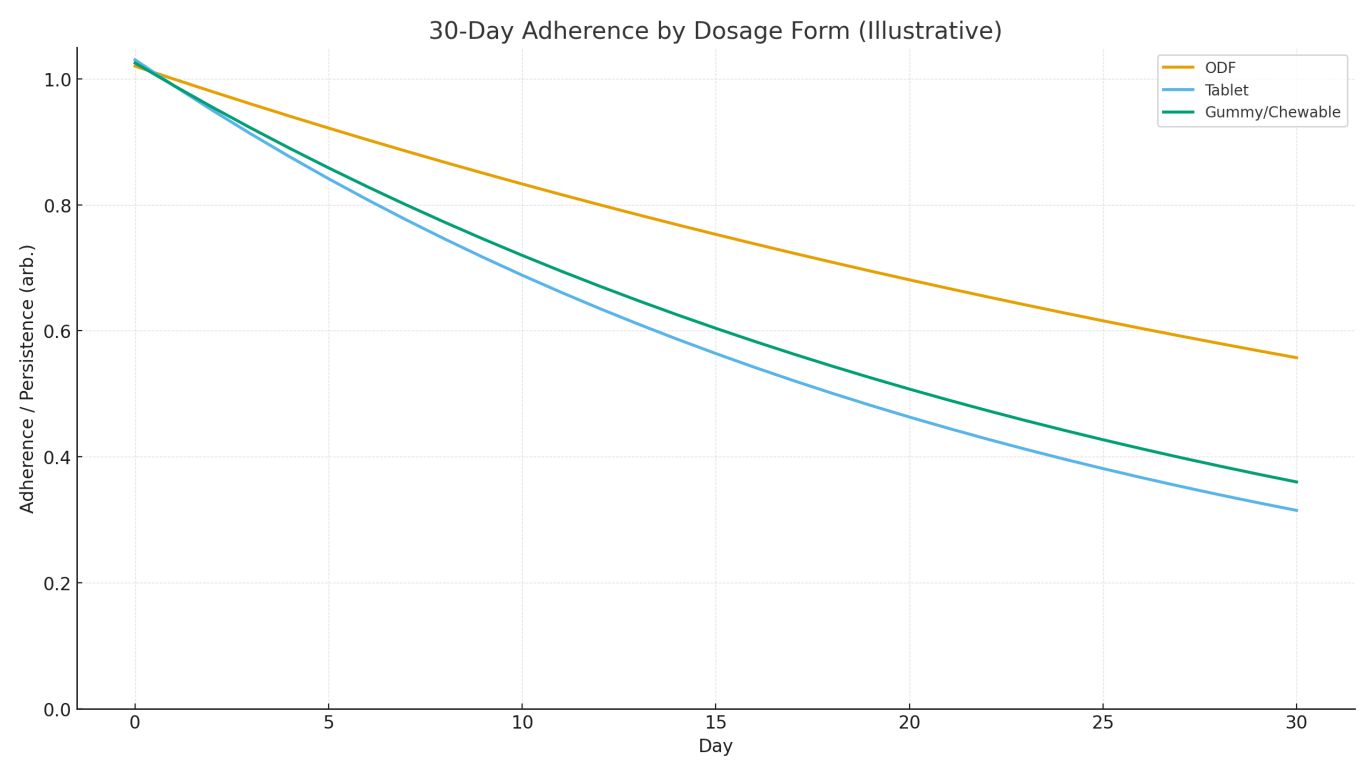
Preferred attributes
Figure 3 demonstrates higher ODF ratings for convenience, swallowability, discreetness, travel-friendliness, with taste competitive after basic masking. These factors correlate with fewer missed doses and higher persistence. [1,6]
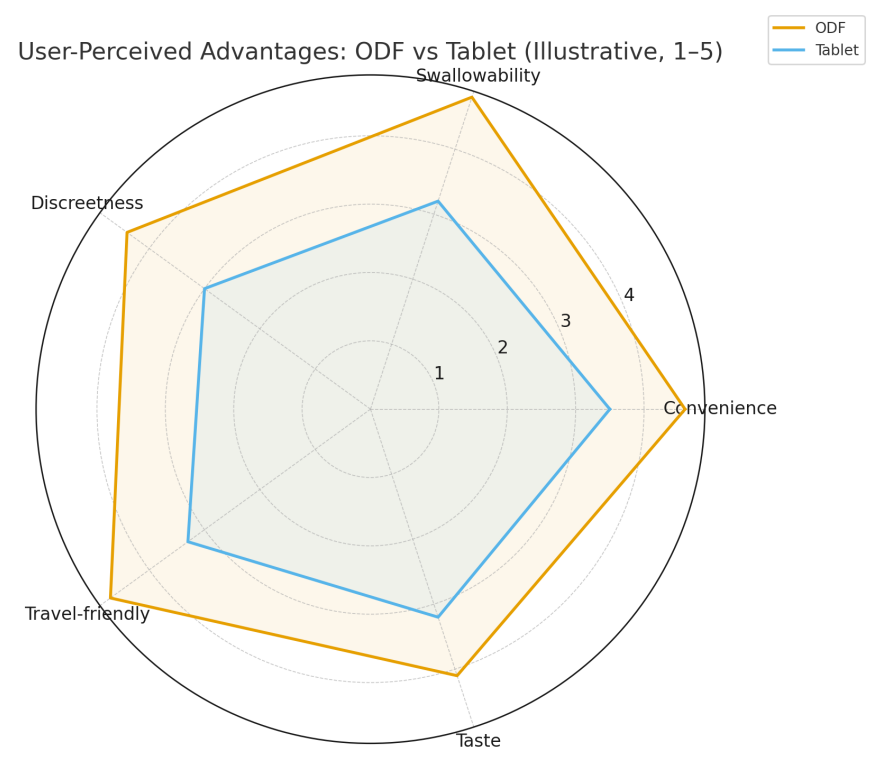
Discussion
Why ODFs can feel faster
ODFs remove tablet disintegration and can deliver some fraction via buccal/sublingual pathways, where feasible. Even when GI absorption dominates, sensory cues of rapid dissolution shape perceived onset. [2–5]
Why ODFs support adherence
No water, discreet, pocketable packs reduce barriers in real-world settings (work, travel, bedtime).
Swallowability advantages matter for elderly, pediatric, and dysphagia populations. [1,6–7]
Quality guardrails to keep advantages real
Maintain (\le 1–2%) thickness CV and robust assay RSD% to ensure dose trust.
Control exit moisture (≈1.6–2.4%) and conditioning RH (≈45–55%) to prevent curl/blocking that would degrade pack opening-force and user experience.
Validate seal windows to avoid “over-seal” (hard to open) and flavor loss from under-barrier laminates. [4–9]
Limitations & when tablets still win
High-dose APIs may exceed practical film loading.
Poorly permeable actives may not gain mucosal advantages; onset gap narrows vs fast-disintegrating tablets.
Sugar-free/label constraints can complicate taste-masking; careful selection of sweetener systems and pH micro-environments remains essential. [3–6,8]
Conclusion
ODFs combine faster perceived onset with higher day-to-day usability, translating into better adherence/persistence—without sacrificing quality when manufactured within defined CPP→CQA windows. For many consumer-health and nutraceutical applications, ODFs represent a next-generation form that aligns pharmacology with human behavior.
References
Adherence science: convenience, swallowability, and persistence in ambulatory care.
Oral mucosal delivery principles: disintegration, dissolution, and transmucosal pathways.
ODF polymers (HPMC/PVA/pullulan) and rheology for rapid dispersion.
QbD/PAT for thin-film coating: inline thickness/moisture/vision, historian/ALCOA+.
Multi-zone drying, exit-moisture, and conditioning for flatness and stability.
Sensory/UX drivers of acceptance: time–intensity, hedonic scoring, usability.
Real-world dosing barriers and dysphagia considerations across age groups.
Packaging integration: laminate OTR/WVTR, seal window and opening-force windows.
Stability and shipping robustness for pouch formats (ACC/RT, vibration/stack tests).
