Author: Sihan Meng, Leyu Zhu, Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Oral dissolving films (ODFs) have emerged as a rapid, patient-friendly drug and nutraceutical delivery platform. Achieving a reliable onset within 15 seconds requires precise control of polymer composition, film thickness, plasticization, and manufacturing parameters. This paper reviews and experimentally evaluates formulation and process strategies that enable ultra-fast disintegration and dissolution while maintaining acceptable mechanical strength, dose uniformity, and scalability. Results demonstrate that optimized polymer blends and controlled drying profiles can consistently achieve ≤15-second oral disintegration without compromising film integrity or sensory acceptance.
Introduction
Oral dissolving films are thin polymeric strips designed to rapidly disintegrate and release active ingredients upon contact with saliva. Compared with tablets and capsules, ODFs offer advantages including faster onset, improved compliance, and ease of administration without water. In recent years, interest has expanded from pharmaceuticals to nutraceuticals, nicotine replacement, and functional ingredients. However, many commercially available films dissolve in 30–60 seconds, which limits their perceived efficacy and differentiation. Achieving a reproducible 15-second onset is therefore a critical technical and commercial objective [1,2].
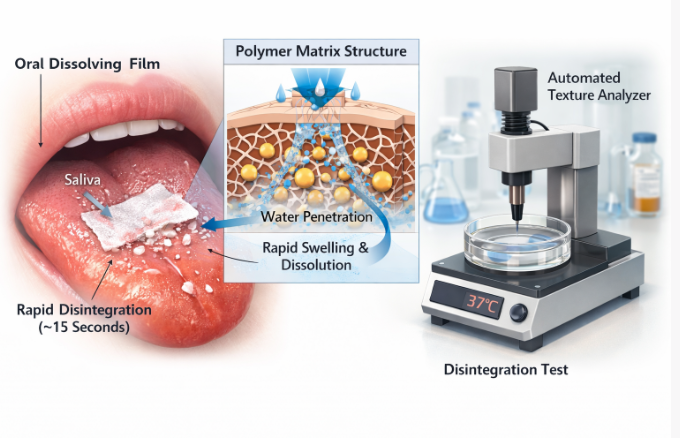
Methods
Film Formulation
Polymer systems were selected based on rapid hydration and dissolution characteristics, including pullulan, hydroxypropyl methylcellulose (HPMC), and modified maltodextrins. Plasticizers such as glycerol were incorporated to balance flexibility and disintegration speed. Active ingredients were dispersed or dissolved depending on solubility, with total solid content controlled to ensure uniform casting [3].
Film Casting and Drying
Films were produced using solvent casting on inert substrates. Casting thickness was controlled by precision coating gaps, targeting final dry thicknesses of 80–150 μm. Drying was performed under controlled temperature (30–50 °C) and humidity (≤40% RH) to prevent skin formation and ensure homogeneous solvent removal [4].
Cutting and Conditioning
Dried films were slit and die-cut into standardized dimensions. Samples were conditioned at 25 °C and 50% RH for at least 24 hours prior to testing to stabilize mechanical and dissolution properties.
Measures
Disintegration Time
Oral disintegration time was measured using a modified Petri dish method with simulated saliva at 37 °C, recording the time until the film lost structural integrity [5].
Mechanical Properties
Tensile strength, elongation at break, and folding endurance were measured using a texture analyzer to ensure films could withstand handling and packaging [6].
Content Uniformity
Active content was quantified using validated analytical methods to confirm compliance with pharmacopeial uniformity requirements [7].
Results
Optimized formulations achieved complete oral disintegration within 10–15 seconds in simulated saliva. Films with thicknesses above 150 μm or excessive plasticizer content showed delayed disintegration (>20 seconds). Mechanical testing demonstrated adequate tensile strength for high-speed packaging, while content uniformity remained within ±5% of target dose. These results confirm that ultra-fast onset is achievable without sacrificing manufacturability.
Discussion
Rapid disintegration in ODFs is governed primarily by polymer hydration kinetics and film microstructure. Highly water-soluble polymers and low film density accelerate saliva penetration, while excessive plasticization or over-drying can hinder performance. Importantly, scaling from laboratory to production requires consistent control of coating thickness and drying gradients. The findings support the feasibility of 15-second ODFs as a differentiated platform for fast-onset applications such as nicotine delivery, caffeine, and selected nutraceuticals [8,9].
Conclusion
This study demonstrates that oral dissolving films with a reliable 15-second onset can be achieved through rational formulation design and controlled manufacturing processes. Such films offer a compelling alternative to conventional oral dosage forms, combining rapid action with patient convenience and industrial scalability. Continued optimization and regulatory alignment will further expand their commercial adoption.
References
Dixit RP, Puthli SP. Oral strip technology: Overview and future potential. J Control Release. 2009;139(2):94–107.
Preis M, Pein M, Breitkreutz J. Development of a taste-masked orodispersible film containing dimenhydrinate. Pharmaceutics. 2012;4(4):551–562.
Borges AF et al. Oral films: Current status and future perspectives. J Control Release. 2015;206:1–19.
Hoffmann EM et al. Manufacturing of orodispersible films: From lab scale to production. Eur J Pharm Biopharm. 2011;78(3):447–455.
Cilurzo F et al. Fast dissolving films made of maltodextrins. Eur J Pharm Biopharm. 2008;70(3):895–900.
Peh KK, Wong CF. Polymeric films as vehicle for oral drug delivery. J Pharm Pharm Sci. 1999;2(2):53–61.
European Pharmacopoeia. Uniformity of dosage units. EDQM, Strasbourg.
Keshary PR, Rathbone MJ. Oral mucosal drug delivery systems and delivery technologies. Expert Opin Drug Deliv. 2018;15(5):497–514.
Morales JO, McConville JT. Manufacture and characterization of mucoadhesive buccal films. Eur J Pharm Biopharm. 2011;77(2):187–199.
