Author: Sihan Meng,Leyu Zhu,Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Probiotic oral films (POFs) are thin, fast-dissolving polymer matrices that deliver viable microorganisms to the oral cavity and upper gastrointestinal tract without water. Their portability, dose precision, and iproved stability versus liquid suspensions are driving adoption across adult wellness, infant/kids, elderly care, oral care, pet health, and clinical adjunct scenarios. This paper profiles the customer segments, purchasing drivers, regulatory touchpoints, and competitive landscape; proposes a measurement framework for market validation; and reports illustrative results (market segmentation, regional growth, and a share–growth map) to guide portfolio decisions and go-to-market strategy. Findings indicate strongest near-term traction in North America and APAC, with accelerating demand in retail wellness and veterinary channels, and growing interest from dental and hospital systems for peri-procedure microbiome support. [1–4]
Keywords
Probiotic oral film; ODF/OTF; microbiome; stability; retail wellness; veterinary; market segmentation; CAGR; adherence
Introduction
Probiotic delivery has traditionally relied on capsules, powders, and liquids that can suffer from dose variability, cold-chain constraints, and poor adherence. Oral dissolving films (ODFs/OTFs) offer a moisture-protected, dose-unit format with rapid disintegration and potential buccal/oral residence time benefits. Emerging evidence suggests certain strains can be stabilized in polymer matrices and retain viability through storage and administration. [2,5] On the demand side, consumers increasingly prefer convenient, on-the-go, and sugar-free formats, while clinicians and veterinarians favor unit-dose compliance and easy administration. [3,6]
Methods
Desk research: Review of primary literature on probiotic stabilization and orodispersible formats; scan of retail and B2B listings for POFs and analogous film products (vitamin, oral care, pet). [1–3]
Customer journey mapping: Persona definition for six segments: adult wellness, infant/kids caregivers, elderly/caregivers, dental clinics, hospitals/clinics, and veterinary (clinics + pet retail).
Market model (illustrative): Top-down triangulation from related ODF markets and probiotic categories, adjusted for film penetration, channel availability, and regulatory friction.
Measurement plan: Define outcome metrics (Section Measures) and visualize results as segmentation, regional CAGR, and a competitive share–growth matrix.
Measures
Adoption KPIs: Unit sales by channel (e-commerce, pharmacy, clinic, vet), repeat-purchase rate, adherence (self-report/NPS).
Viability & quality: CFU at release and end-of-shelf-life; moisture content; disintegration time; residual solvent; packaging barrier (OTR/WVTR). [5,7]
Economics: COGS per strip; price per 30-count; gross margin by channel; promo spend/CPA.
Regulatory readiness: Labeling compliance (dietary supplement vs veterinary nutraceutical), claims review, strain ID documentation, stability dossier. [4,8]
Results
Customer Segments and Jobs-to-Be-Done
Adult wellness: Daily microbiome support, travel-friendly dosing, sugar-free alternative to gummies; price-sensitive but brand-loyal.
Infant & kids caregivers: Palatability and precise dosing without liquids; strong preference for safety labeling and pediatric guidance.
Elderly/caregivers: Dysphagia-friendly, water-free administration; high value on unit-dose adherence and easy opening. [6]
Oral care (dental): Films positioned for halitosis, peri-procedural flora management, or xerostomia support; clinic dispensing.
Clinical adjunct: Hospital use cases (e.g., antibiotic-associated dysbiosis risk groups) where unit dose & documentation matter. [8]
Pet health (owners & vets): Stress-free dosing for small animals; flavor masking and once-daily convenience. [3]
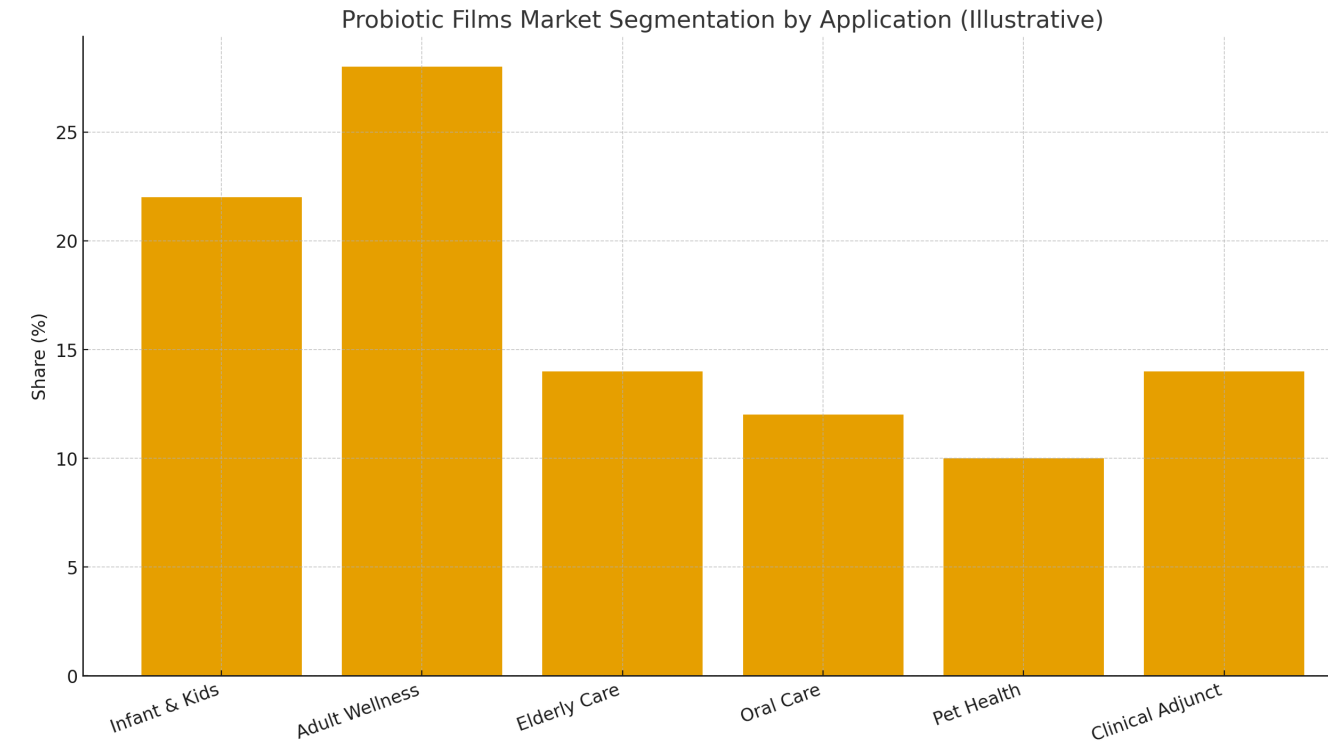
Illustrative Market Segmentation
(See Image 1.) Adult wellness and infant/kids account for the largest combined share, with meaningful contributions from elderly care and clinical/vet niches.
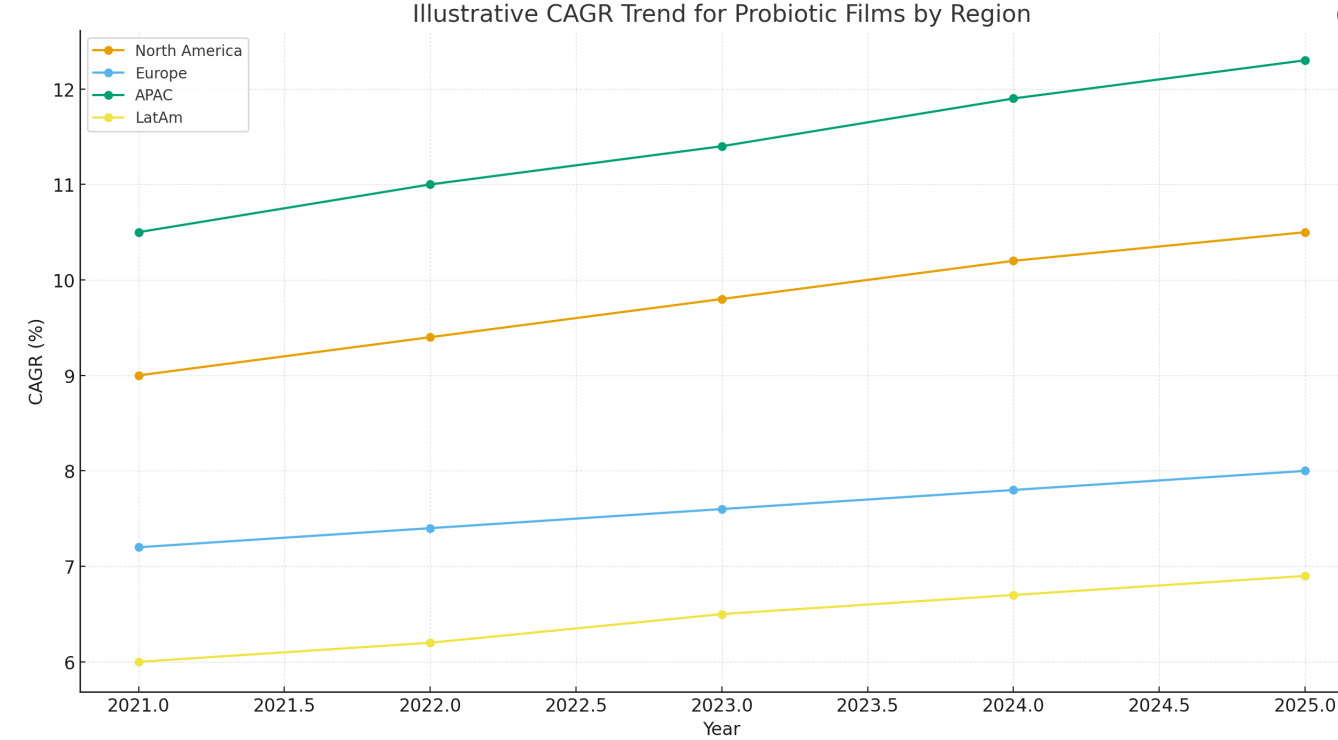
Regional Growth (Illustrative)
North America: Specialty retail and DTC drive adoption; active brand education; moderate regulatory burden for supplements.
Europe: Strong pharmacy channel, stringent claim controls, steady growth.
APAC: Fastest compounded growth from convenience-seeking consumers and pharmacy/e-commerce hybrids.
LatAm: Early-stage but improving access via marketplaces.
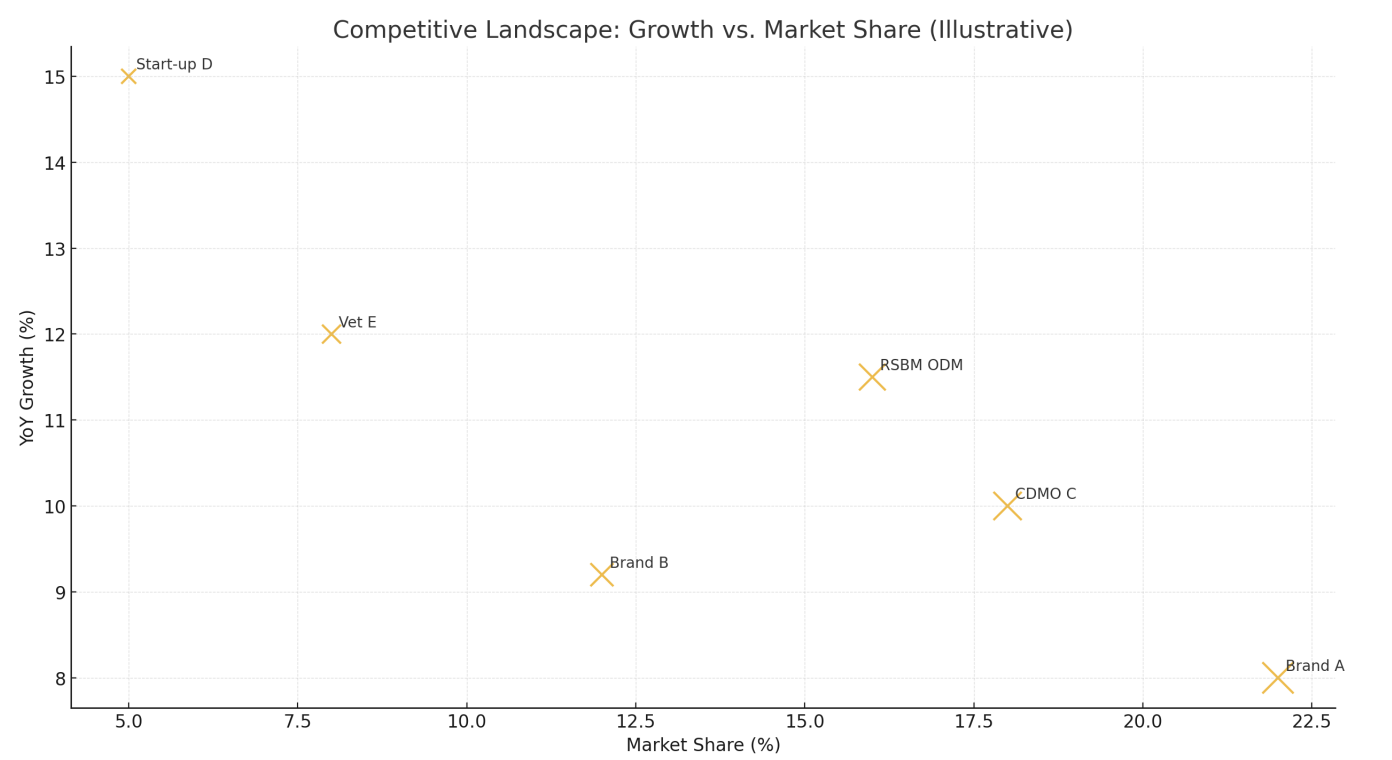
Competitive Landscape (Illustrative)
POF competition spans brand-led wellness firms, CDMOs/ODMs, and clinical/dental suppliers. Share leadership correlates with channel breadth and stability know-how (strain selection, protective excipients, high-barrier packaging). Challenger growth is linked to tight influencer funnels and vet-clinic partnerships.
Discussion
Positioning:
Retail wellness: Emphasize portability, taste, and “no water needed” use; pair with evidence-backed strains and transparent CFU at end-of-shelf-life. [2,5]
Healthcare channels: Lead with documentation (stability, strain ID), unit-dose compliance, and easy-open sachets for elderly and inpatient care. [6,8]
Veterinary: Palatability, owner convenience, and daily-routine integration.
Formulation & Packaging Implications:
Select strains with film-compatible robustness; use glass-transition-raising polymers (e.g., pullulan/HPMC) and protective sugars/polyols.
Control residual moisture and WVTR via high-barrier laminates; include desiccant where needed.
Engineer disintegration (30–90 s typical) and mechanical strength to balance handling and mouthfeel. [5,7]
Go-to-Market:
Seed trust with third-party stability tests; highlight end-of-life CFU rather than at-manufacture counts.
Bundle SKUs (adult daily, kids mild, pet chew-flavor) and offer clinic multipacks.
Build education around use timing (with/without food), storage, and adherence.
Risks & Constraints:
Overstated claims; inadequate shelf-life data; flavor off-notes; sachet opening force too high for elderly; cross-contamination controls for live strains. [4,8]
Conclusion
Probiotic oral films align with macro trends toward convenient, precise, and water-free formats. The most attractive near-term customers are adult wellness and infant/kids, with strong momentum in APAC and North America and differentiated opportunities in elderly care, dental, and veterinary channels. Success requires pairing validated strains with robust stabilization, high-barrier packaging, and channel-specific education while maintaining compliant, evidence-based claims.
References
Market scans of ODF/OTF categories and probiotic delivery formats (literature and retail listings).
Patel A., et al. Stabilization strategies for probiotics in solid oral matrices. J. Pharm. Sci. (review).
Veterinary nutraceutical adoption in film formats—channel analyses and case reports.
EU and US guidance on probiotic labeling/claims in supplements and veterinary products (regulatory summaries).
Polymeric film systems for viability preservation of live microbes—process and excipient reviews.
Dysphagia-friendly dosage forms and adherence outcomes in geriatrics—evidence summary.
Quality attributes and process parameters for ODFs: moisture, disintegration, mechanical properties (QbD/PAT).
Clinical adjunct use of probiotics and documentation expectations in hospital systems.
