Author: Sihan Meng, Leyu Zhu, Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Packaging selection is a decisive factor in the performance, stability, compliance, and commercial viability of oral dissolving film (ODF) products. Beyond containment, packaging determines moisture protection, dose integrity, user behavior, regulatory acceptance, and manufacturing cost. This paper provides a systematic comparison of four widely used ODF packaging formats—sachets, blisters, pouches, and boxes—analyzing their technical advantages and limitations across pharmaceutical, nutraceutical, and regulated consumer applications. By aligning packaging choice with product characteristics and use scenarios, this study offers a practical framework to support rational ODF packaging decisions from development to commercialization.
Introduction
Oral dissolving films are inherently sensitive to environmental factors, particularly moisture, mechanical stress, and repeated handling. While formulation and process optimization are essential, inappropriate packaging can negate these efforts by introducing instability, dosing errors, or poor user experience [1].
In practice, packaging decisions are often driven by cost or equipment availability rather than by product requirements and use scenarios. Such mismatches frequently lead to downstream issues, including brittleness, blocking, overdosing risk, or regulatory challenges [2]. This paper focuses on four common ODF packaging formats—sachets, blisters, pouches, and boxes—and examines how each format influences product performance and suitability in different contexts.
Methods
The study employed a comparative, scenario-based evaluation approach:
Packaging Format Definition
Sachets, blisters, pouches, and boxes were defined according to sealing method, access mode, and unit-dose control.Scenario Mapping
Typical application scenarios were identified, including clinical dosing, daily supplementation, on-the-go use, and professional or institutional settings.Risk–Benefit Assessment
Each format was assessed for moisture protection, dose accuracy, user compliance, manufacturability, and cost.Manufacturing Compatibility Review
Compatibility with high-speed ODF lines, pouching/blistering equipment, and secondary packaging operations was evaluated.
Measures
The following indicators were used to compare packaging formats:
Moisture Barrier Performance: Resistance to water vapor ingress during storage and use.
Dose Integrity: Ability to ensure single-dose access and prevent multi-strip extraction.
User Compliance: Ease of correct and repeatable use.
Operational Complexity: Equipment requirements, line speed, and scrap rate.
Regulatory Suitability: Alignment with pharmaceutical and regulated consumer product expectations.
These measures reflect both technical and commercial considerations [3].
Results
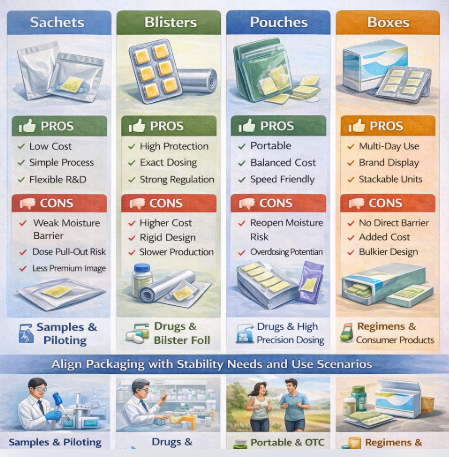
1. Sachets
Sachets typically contain one or multiple films in a simple sealed envelope.
Advantages:
Low material and equipment cost
Flexible for pilot production and sampling
Simple sealing process
Limitations:
Limited moisture barrier unless high-grade laminates are used
Risk of multi-dose access when multiple films are packed together
Less premium user perception
Sachets performed best in short-term use, sampling, and cost-sensitive applications.
2. Blisters
Blisters provide individual cavities for each film, usually sealed with aluminum or composite lidding.
Advantages:
Excellent moisture and oxygen protection
Precise unit-dose control
Strong regulatory acceptance
Limitations:
Higher tooling and material costs
Reduced flexibility for frequent design changes
Slower line speeds compared with pouching
Blisters were consistently superior for pharmaceutical and high-risk dosing scenarios.
3. Pouches
Pouches are flexible packages that may contain one or multiple films.
Advantages:
Good balance between cost and protection
Compatible with high-speed packaging lines
Suitable for portable and on-the-go use
Limitations:
Repeated opening increases moisture exposure for multi-dose pouches
User discipline required to avoid overdosing
Pouches showed optimal performance in consumer-facing and premium nutraceutical products.
4. Boxes
Boxes are secondary packaging formats that typically contain sachets, blisters, or pouches.
Advantages:
Improved branding and information display
Protection during transport and storage
Suitable for multi-day or multi-week regimens
Limitations:
No direct moisture protection without primary packaging
Additional cost and packaging steps
Boxes function primarily as a supportive, rather than protective, packaging layer.
Discussion
The comparative analysis demonstrates that no single packaging format is universally optimal for ODF products. Instead, packaging must be selected based on use scenario, dosing risk, and environmental exposure rather than cost alone. Blisters offer maximum control and protection, making them ideal for pharmaceutical applications. Sachets and pouches provide flexibility and cost efficiency but require careful moisture management. Boxes enhance usability and branding but cannot substitute for effective primary packaging [4].
Critically, packaging selection should be integrated early in development, as it directly influences film thickness, moisture targets, and mechanical strength requirements.
Conclusion
ODF packaging is a strategic design choice that directly impacts stability, compliance, and user experience. Sachets, blisters, pouches, and boxes each serve distinct roles depending on application and regulatory context. By systematically evaluating the pros and cons of each format and aligning them with intended use scenarios, manufacturers can reduce development risk, improve product reliability, and support successful commercialization.
References
Borges, A. F., Silva, C., Coelho, J. F. J., & Simões, S. (2015). Oral films: Current status and future perspectives. Journal of Controlled Release, 206, 1–19.
Preis, M., Knop, K., & Breitkreutz, J. (2013). Mechanical strength test for orodispersible films. International Journal of Pharmaceutics, 461(1–2), 22–29.
Dixit, R. P., & Puthli, S. P. (2009). Oral strip technology: Overview and future potential. Journal of Controlled Release, 139(2), 94–107.
Cilurzo, F., Musazzi, U. M., Franzé, S., Selmin, F., & Minghetti, P. (2016). Orodispersible dosage forms: Biopharmaceutical improvements and regulatory requirements. Drug Discovery Today, 21(5), 707–716.
