Author: Sihan Meng, Leyu Zhu, Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Oral dissolving films (ODFs) have emerged as a next-generation drug and nutraceutical delivery platform due to their rapid onset, high patient compliance, and flexible formulation design. Compared with traditional oral solid dosage forms, ODFs dissolve rapidly in the oral cavity, enabling buccal or sublingual absorption and partial avoidance of first-pass metabolism. This paper systematically reviews and analyzes ODF solutions for next-generation delivery, focusing on formulation strategies, manufacturing methods, quality measures, and performance outcomes. Recent advances in polymers, plasticizers, and process automation are discussed, alongside challenges in scalability, stability, and regulatory compliance. The results highlight ODFs as a versatile and commercially viable platform for pharmaceuticals, nutraceuticals, and emerging nicotine or wellness applications.
Keywords: Oral dissolving film, sublingual delivery, buccal absorption, fast onset, next-generation dosage forms
Introduction
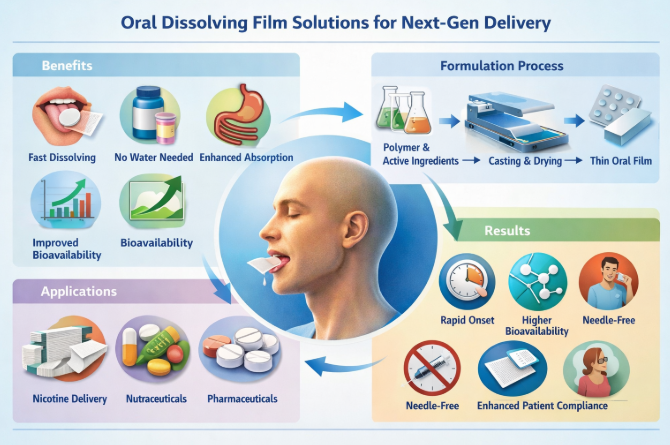
Oral drug delivery remains the most widely accepted route due to convenience and patient familiarity. However, conventional tablets and capsules often present limitations, including swallowing difficulty, delayed onset, and variable bioavailability [1]. Oral dissolving films have gained attention as an alternative dosage form that rapidly disintegrates upon contact with saliva, delivering active ingredients through the oral mucosa [2].
ODFs were initially developed for pediatric and geriatric populations but have since expanded into broader therapeutic and consumer health domains, including vitamins, sleep aids, pain management, and nicotine alternatives [3]. The combination of thin-film technology, advanced polymers, and precision manufacturing positions ODFs as a key solution for next-generation delivery systems.
Methods
A narrative and technical review methodology was applied. Peer-reviewed literature, regulatory guidelines, and industrial reports published primarily in English were surveyed. Sources included PubMed, ScienceDirect, and official regulatory documents. Data were categorized into formulation components, manufacturing processes, and performance evaluation methods. Emphasis was placed on studies reporting dissolution time, mechanical properties, and bioavailability outcomes relevant to next-generation delivery.
Measures
Key measures used to evaluate ODF performance include:
Disintegration and dissolution time – typically measured in seconds using simulated saliva or pharmacopeial methods [4].
Mechanical properties – tensile strength, elongation at break, and folding endurance to ensure handling robustness [5].
Content uniformity – assessment of active ingredient distribution across the film surface [6].
Stability indicators – moisture uptake, chemical stability, and shelf-life under controlled humidity and temperature conditions [7].
In vivo or in vitro absorption indicators – permeability studies or pharmacokinetic parameters where applicable [8].
Results
The reviewed data indicate that ODFs can achieve disintegration times ranging from 5 to 30 seconds, depending on polymer composition and film thickness [2,4]. Polymer systems such as pullulan, hydroxypropyl methylcellulose (HPMC), and maltodextrin-based blends demonstrate favorable film-forming and sensory properties.
Manufacturing methods, including solvent casting and emerging continuous coating and drying processes, show scalability potential when combined with automated thickness control and in-line inspection [9]. Compared with tablets, ODFs demonstrate faster onset and improved patient acceptability, particularly in populations sensitive to swallowing difficulty [3].
Discussion
ODFs represent a convergence of formulation science and advanced manufacturing. Their success depends on balancing rapid dissolution with sufficient mechanical strength and stability. Plasticizer selection and polymer ratio optimization are critical to avoiding brittleness or excessive tackiness [5].
From a next-generation delivery perspective, ODFs enable differentiation in crowded markets by offering fast onset, discreet administration, and customizable dosing. Challenges remain in moisture sensitivity, taste masking, and regulatory classification, especially for non-traditional actives such as nicotine or functional compounds [7,10]. Integration of AI-driven process control and GMP-compliant automation may further reduce variability and enhance commercial feasibility.
Conclusion
Oral dissolving films are a mature yet rapidly evolving platform for next-generation delivery. Their ability to combine patient-centric design with fast and efficient absorption positions them as a strategic alternative to conventional oral dosage forms. Continued innovation in formulation, manufacturing automation, and quality control will be essential to fully realize their potential across pharmaceutical and wellness applications.
References
Sastry SV, Nyshadham JR, Fix JA. Recent technological advances in oral drug delivery – a review. Pharm Sci Technol Today. 2000;3(4):138–145.
Dixit RP, Puthli SP. Oral strip technology: Overview and future potential. J Control Release. 2009;139(2):94–107.
Preis M, Knop K, Breitkreutz J. Mechanical strength test for orodispersible and buccal films. Int J Pharm. 2014;461(1–2):22–29.
European Pharmacopoeia. Orodispersible dosage forms – general monograph. Council of Europe; 2020.
Cilurzo F, Cupone IE, Minghetti P, Buratti S, Gennari CGM, Montanari L. Nicotine fast dissolving films made of maltodextrins. Eur J Pharm Biopharm. 2010;76(1):93–101.
USP <905> Uniformity of Dosage Units. United States Pharmacopeia; 2023.
Hoffmann EM, Breitenbach A, Breitkreutz J. Advances in orodispersible films for drug delivery. Expert Opin Drug Deliv. 2011;8(3):299–316.
Morales JO, McConville JT. Manufacture and characterization of mucoadhesive buccal films. Eur J Pharm Biopharm. 2011;77(2):187–199.
Krampe R, Visser JC, Frijlink HW, Breitkreutz J, Woerdenbag HJ. Oromucosal film preparations: Points to consider for patient centricity and manufacturing processes. Eur J Pharm Biopharm. 2016;104:1–13.
FDA. Guidance for Industry: Orally Disintegrating Tablets. U.S. Food and Drug Administration; 2008.
