What Is an Oral Disintegrating Film (ODF/OTF): Definition, Structure, and Mechanism of Action
Author: Sihan Meng,Leyu Zhu,Pengcheng Shi
Affiliation: RSBM
Email: pengchengshi@biotechrs.com; pcspc9@gmail.com
Abstract
Oral Disintegrating Films (ODF), also referred to as Oral Thin Films (OTF), are rapidly dissolving polymeric dosage forms designed for administration on the tongue or buccal mucosa without the need for water. Over the past decade, ODFs have gained increasing attention as an alternative to conventional tablets, capsules, and liquids, particularly for pediatric, geriatric, and dysphagic populations. This paper systematically defines ODFs, analyzes their structural components, and explains their mechanisms of action from disintegration to drug absorption. Manufacturing methods, performance measures, and formulation outcomes are reviewed to provide a comprehensive understanding of this dosage form. The discussion highlights advantages, limitations, and future development directions for pharmaceutical, nutraceutical, and nicotine-related applications.
Keywords: Oral Disintegrating Film, Oral Thin Film, Fast Dissolving Film, Buccal Delivery, Drug Delivery System
Introduction
Oral solid dosage forms dominate global pharmaceutical markets; however, they present challenges such as swallowing difficulty, delayed onset of action, and reduced patient compliance in specific populations [1]. To overcome these limitations, alternative oral delivery systems have been developed, among which Oral Disintegrating Films (ODF/OTF) represent a rapidly expanding platform.
ODFs are ultra-thin polymeric films that disintegrate within seconds upon contact with saliva, releasing active ingredients directly into the oral cavity [2]. Compared with orally disintegrating tablets (ODTs), ODFs offer superior dosing accuracy, improved mouthfeel, faster disintegration, and greater formulation flexibility [3]. Their applications now extend beyond pharmaceuticals to nutraceuticals, functional supplements, and nicotine replacement therapies.
Definition of Oral Disintegrating Film
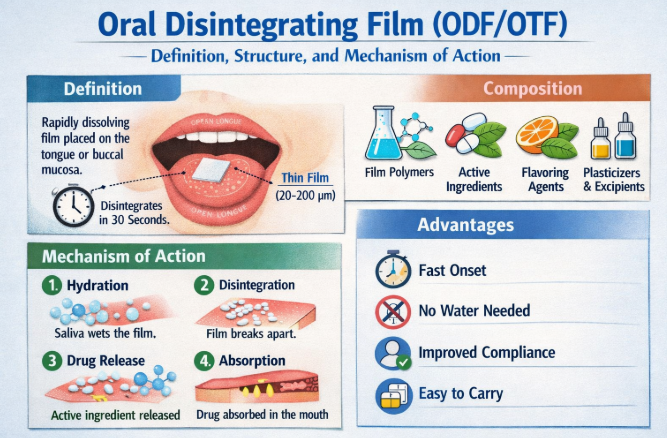
An Oral Disintegrating Film is a solid dosage form consisting of one or more layers of hydrophilic polymers incorporating active pharmaceutical or functional ingredients. When placed on the tongue or buccal mucosa, the film hydrates, disintegrates, and releases its payload rapidly, typically within 5–60 seconds, without requiring water [4].
Key defining characteristics include:
Film thickness typically ranging from 20 to 200 μm
Rapid disintegration in saliva
Accurate unit-dose delivery
Direct oral or transmucosal administration
Structural Composition of ODF
The structure of an ODF is engineered to balance mechanical strength, rapid dissolution, and palatability. A typical ODF consists of the following components [5,6]:
1. Film-Forming Polymers
These form the backbone of the film and determine its mechanical and dissolution properties. Common examples include pullulan, hydroxypropyl methylcellulose (HPMC), polyvinyl alcohol (PVA), and maltodextrins.
2. Active Ingredients
Active components may include pharmaceuticals, vitamins, minerals, botanical extracts, caffeine, melatonin, or nicotine. Dose uniformity is critical due to the low mass of the film.
3. Plasticizers
Plasticizers such as glycerol, polyethylene glycol, or propylene glycol enhance flexibility and prevent brittleness.
4. Flavoring and Sweetening Agents
Taste masking is essential due to prolonged oral residence. Sweeteners and flavors improve patient acceptability.
5. Functional Excipients
Surfactants, saliva stimulants, coloring agents, and stabilizers may be incorporated to optimize performance.
Methods
ODFs are commonly manufactured using solvent casting, hot-melt extrusion, or semi-solid casting techniques [7].
Solvent Casting (Most Common)
Preparation of polymer solution
Incorporation of active ingredients and excipients
Casting onto a flat substrate
Controlled drying
Cutting and packaging
This method offers excellent content uniformity and scalability.
Measures
The quality and performance of ODFs are evaluated using standardized pharmaceutical tests [8,9]:
Film thickness and weight variation
Tensile strength and elongation
Disintegration time
Dissolution profile
Content uniformity
Moisture uptake and stability
These parameters ensure consistency, safety, and efficacy.
Results
Studies consistently demonstrate that ODFs disintegrate faster than conventional oral dosage forms, often within 30 seconds [10]. In vitro and in vivo evaluations show rapid release of active ingredients and, in some cases, improved bioavailability due to partial buccal absorption and avoidance of first-pass metabolism [11].
Patient preference studies report higher acceptance rates compared with tablets, especially among children and elderly patients [12].
Mechanism of Action
The mechanism of action of ODFs involves several sequential steps:
Hydration: Saliva penetrates the hydrophilic polymer matrix.
Swelling and Disintegration: The film softens and breaks apart rapidly.
Drug Release: Active ingredients are released into the oral cavity.
Absorption:
Local absorption through oral mucosa (buccal/sublingual)
Swallowing of dissolved drug for gastrointestinal absorption
This dual absorption pathway enables both rapid onset and systemic delivery [13].
Discussion
ODFs combine the convenience of solid dosage forms with the rapid action of liquid formulations. Their advantages include portability, precise dosing, improved compliance, and suitability for water-free administration. However, challenges remain, such as limited drug loading capacity, sensitivity to moisture, and formulation complexity for bitter or high-dose actives [14].
Emerging technologies, including multilayer films, nanoparticle incorporation, and resin-based taste masking, are expanding the applicability of ODF platforms [15].
Conclusion
Oral Disintegrating Films represent a versatile and patient-friendly drug delivery system with broad applications across pharmaceutical and functional product categories. Understanding their definition, structure, and mechanism of action is essential for rational formulation design and successful commercialization. With continued advancements in materials science and manufacturing technology, ODFs are poised to become a mainstream oral dosage form in global markets.
References
Fu Y et al. Expert Opin Drug Deliv. 2004;1(4):673–690.
Dixit RP, Puthli SP. J Control Release. 2009;139(2):94–107.
Preis M. J Pharm Pharmacol. 2013;65(2):157–170.
European Pharmacopoeia. Oromucosal preparations.
Bala R et al. Int J Pharm Investig. 2013;3(2):67–76.
Morales JO, McConville JT. Ther Deliv. 2011;2(5):637–646.
Cilurzo F et al. Eur J Pharm Biopharm. 2008;70(3):895–900.
USP <701> Disintegration Test.
USP <905> Uniformity of Dosage Units.
Arya A et al. Int J PharmTech Res. 2010;2(1):576–583.
Hoffmann EM et al. Pharm Res. 2011;28(8):1914–1922.
Borges AF et al. Int J Pharm. 2015;494(1):332–339.
Shojaei AH. J Pharm Pharm Sci. 1998;1(1):15–30.
Keshari R, Keshari S. J Drug Deliv Ther. 2014;4(4):1–7.
Preis M et al. Drug Dev Ind Pharm. 2014;40(2):152–160.
