By 2024, the global market is already a multi-billion-dollar business:
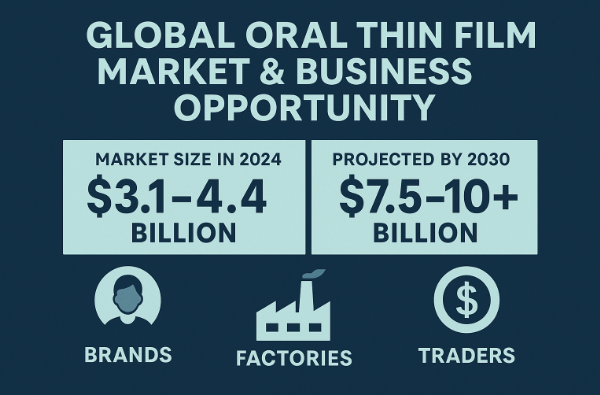
Many market reports estimate the global oral thin film market size in 2024 at around USD 3.1–4.4 billion.
Projections to 2030–2035 generally fall in the USD 7.5–10+ billion range.
The expected compound annual growth rate (CAGR) is roughly 8%–11% in the coming years.
Different agencies use different methodologies, but they share one clear view:
this is not a tiny niche anymore. It is a fast-growing global market, driven by:
ageing populations and swallowing difficulties,
people who dislike pills,
and consumers who want convenient, good-looking, “techy” dosage forms.
If you are looking at oral dissolving film / oral thin film / orodispersible film as a business, you usually have three questions:
Can this product really make money?
Is it suitable for my role (brand owner / factory / foreign-trade seller)?
What does the full process from “raw material” to “finished product” actually look like?
The good news: oral film products are already being used in sleep, energy, beauty, breath freshening, sexual health, pet nutrition and many other categories. For you, this means:
Brand owners – use a new dosage form + better design to grab consumer attention and increase margin.
Factory owners – use one ODF production line to serve many brands and SKUs, maximising utilisation.
Foreign-trade newcomers – act as a “project manager” type integrator, connecting overseas clients with ODF OEM/ODM resources and getting paid for your expertise.
This article uses a text-based flowchart and five “golden steps” to explain:
How each of the three roles makes money.
The 10 key steps from raw materials to finished oral film.
Which steps most strongly affect your profit and bargaining power.
Concrete action tips for brands, factories and foreign-trade sellers.
A FAQ that answers the most common questions in one go.
I. First, understand how each role makes money
1.1 Brand owners: use a new dosage form to win mindshare and raise margins
Your strengths: channels, users, brand.
What oral dissolving film can do for you:
Turn existing functions into oral dissolving film versions:
Melatonin oral film (sleep)
Vitamin B complex oral film
Caffeine energy oral strip
Beauty / collagen oral film
Build stronger selling points:
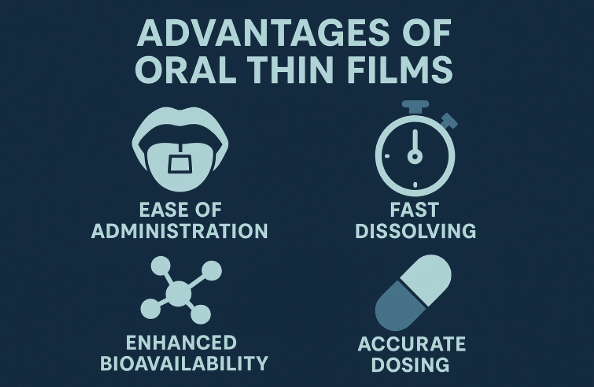
“Dissolves in 3 seconds, no swallowing, no water needed”
“Pocket-sized, for metro, office, travel”
“Ideal for elderly, children and people who hate tablets”
Profit space comes from:
Different dosage form → supports higher unit price.
“New product” label → supports higher margin.
Functional combinations → bundles, co-branding, limited editions.
1.2 Factory owners: sell a full “formulation + line + packaging” solution
Your core assets: equipment, workshops, technical team.
For a factory, oral film is valuable because one ODF production line can serve many brands and SKUs:
You are not just selling a line, you are selling a full package:
Formulation development
Lab / pilot / scale-up production
Packaging material selection and design
Mass production + stable supply
Revenue can be broken into:
Formulation development fees (one-off or milestone-based)
Processing fees (priced per 10,000 or 100,000 films)
Packaging material spread (film, sachets, cartons)
Long-term repeat orders (true profit comes from continuity, not one-shot “big kill”)
1.3 Foreign-trade newcomers: become a project-manager-type ODF integrator
You may not own a factory, but you can own:
A clear understanding of the ODF manufacturing process.
Reliable contract manufacturers.
A set of professional explanations that convince overseas buyers.
Your main job is not running the line yourself, but:
Translating buyer needs into executable projects:
Functional goal (sleep / energy / beauty / pet)
Dose, film size, flavour, packaging
Regulatory and labelling requirements in the target market
Linking all parties:
Factory → packaging → logistics → documentation
Your core value = you understand the process and risk points better than a “pure price sender”.
Whenever a buyer asks:
“How do you produce this?”
you can walk them through a “from raw materials to finished product” flow from left to right. That’s what wins you projects.
II. The complete process from raw materials to finished oral film (text flowchart)
Overall process:
Raw material preparation → Solution / slurry preparation → Filtration & deaeration → Coating → Drying & thickness control → Cooling & rewinding → Slitting → Punching (die-cutting) → Counting & packaging → Final QC & shipping
Along the way you can naturally use SEO keywords such as
oral dissolving film, oral thin film, orodispersible film, ODF production line, ODF OEM/ODM, oral film machine, etc., where appropriate.
2.1 Raw material preparation
Film-forming polymers
e.g. HPMC, PVA, Pullulan
Active ingredients
Vitamins, melatonin, caffeine, herbal extracts, pet nutrition ingredients, etc.
Excipients
Sweeteners, flavours, plasticisers (glycerin, PEG, etc.), stabilisers, colours
Core point: this part defines what the product is and for whom.
2.2 Solution / slurry preparation
Add purified water + film-forming polymer into a mixing tank, controlling speed and temperature.
Add actives, sweeteners, flavours, plasticisers in the required order.
Monitor:
Viscosity
Solid content
pH
For clients, you can explain:
“This stage determines taste, disintegration speed and stability. It’s the core formulation step of any oral film.”
2.3 Filtration & deaeration
Use filters or cartridges to remove particles and lumps.
Vacuum deaeration to reduce bubbles and pinholes and to avoid non-uniform film.
Often used as a “professional vs. backyard workshop” indicator:
Do you have deaeration equipment?
Do you use standardised filter cartridges?
2.4 Coating
Use a slot-die coater or comma coater to apply the solution evenly onto a moving web (usually a PET process belt).
Key parameters:
Coating speed (m/min)
Coating weight (g/m²)
Coated width and edge neatness
For buyers you can emphasise:
“We use a continuous ODF coating-drying line, suitable for large-scale production.”
2.5 Drying & thickness control
Multi-zone drying ovens with a temperature profile defined by the process.
Online thickness measurement to monitor the film thickness in real time and adjust automatically.
Targets:
Reduce moisture to a safe range for stability and packaging.
Avoid degrading the active ingredients.
2.6 Cooling & rewinding
After drying, first cool the film to avoid softness and sticking.
Then rewind into large rolls under controlled tension.
This step is the foundation for stable downstream slitting.
2.7 Slitting
Slit the large roll into multiple narrow rolls according to target film size.
Carefully control tension and edge alignment to minimise rough edges and “snaking”.
To buyers you can say:
“This is the transition from ‘wide web’ to ‘near-final size’.”
2.8 Punching / die-cutting
Use die-cutting machines to punch the narrow rolls into individual films:
Strips, squares, rounded corners, etc.
Different shapes for human vs. pet products if needed.
Dimensional accuracy and edge quality directly affect appearance and user experience.
2.9 Counting & packaging
Typical packaging formats:
Single-film sachets
Multi-strip cartons
Pocket-size plastic dispensers (e.g. Listerine-style)
An oral film machine or packaging line usually integrates:
Automatic counting
Automatic sachet or case filling
Sealing, coding, labelling, etc.
2.10 Final QC & shipping
Typical tests include:
Appearance, dimensions, weight variation
Content uniformity and total content
Disintegration / dissolution time
Microbial limits
Stability studies as required
To summarise for external communication, you can say:
“From raw material receipt to finished goods dispatch, every batch of oral thin film is fully traceable with complete QC records.”
III. The steps that most affect how much money you can make
3.1 Does formulation and positioning match the target customer?
High ticket price categories: beauty, skin brightening, sexual health, mood support, etc.
→ more emphasis on experience and packaging.Mass-volume categories: vitamins and basic supplements.
→ more emphasis on cost control and capacity.Formulation must balance:
Taste masking
Disintegration time
Mechanical properties (strength and flexibility)
3.2 Stability of coating–drying–thickness control
Thickness variation = non-uniform API content = big buyers walk away.
For factories, this directly decides:
Whether you can enter high-standard markets (US / EU / Japan).
Whether you can present solid test reports and process consistency.
3.3 Does your packaging help tell the story?
Premium route:
Individual sachets + high-end printed cartons + scenario-based copy.
Functional route:
Emphasise dose, speed of disintegration and safe ingredients.
Factories and traders can create a “packaging menu” to help clients decide quickly.
3.4 Lead time and minimum order quantity (MOQ)
Many buyers are blocked by MOQ alone.
You can design:
A “development MOQ” for testing and crowdfunding.
A “mass production MOQ” for long-term brand building.
This tiered approach itself is a business and product innovation.
IV. Practical tips for each of the three roles
4.1 For brand owners: start with one or two pilot products
Choose functions that consumers understand immediately:
Sleep (melatonin oral film)
Energy (caffeine energy strip)
Breath freshening (breath freshening oral film)
Highlight these phrases on listings and packaging:
fast dissolving,no water needed,pocket-size,sugar freeTreat oral film as an upgraded dosage form, not just “one more SKU”.
4.2 For factory owners: use the full-process flowchart as your credibility tool
Turn the 10-step process into:
A one-page section on your website with simple diagrams.
A one-page bilingual PDF for WhatsApp / email.
Use consistent wording on your website, Alibaba and LinkedIn:
ODF OEM/ODM full processfrom formula to finished oral filmoral thin film production line
4.3 For foreign-trade newcomers: become an “ODF project manager”
Be able to explain the 10 steps smoothly in English.
When working with factories, always ask:
Which actives can you handle?
What existing film sizes / moulds do you have?
What test reports and certifications can you provide?
When talking to buyers, emphasise your value:
You help them screen factories, control risk, coordinate communication and documents, instead of only forwarding a price.
V. An action checklist you can use immediately
Decide your role: brand owner, factory or foreign-trade seller.
Pick one easy entry scenario: sleep, energy, beauty, breath freshening or pet products.
Draw your own 10-step flowchart based on this article:
“Raw materials” on the left, “Finished packaging” on the right.
10 nodes in the middle, each with 1–2 lines of explanation.
Place this flowchart on your external touchpoints:
Website, Alibaba listing, PPT, product PDF.
Next time you talk to a client, explain through “process + scenario”, not just a FOB price.
FAQ: Common questions about the ODF process from raw materials to finished product
Q1: What is a typical MOQ for oral dissolving film?
A: It depends on formulation, packaging and target market. For health-type products, tens of thousands of films per SKU is common. When both formulation and packaging are fully customised, MOQ is usually higher. You can negotiate a “trial MOQ” and a “mass-production MOQ” with the factory.
Q2: How long does it take from samples to bulk production?
A: If the formulation is similar to existing ones, lab samples can be ready in about 1–2 weeks, and bulk production in about 3–5 weeks. Projects involving registration, special testing or complex regulations will take longer overall.
Q3: How does the per-film cost compare with tablets or capsules?
A: Raw material cost is not always much higher, but the process and packaging require more precision. The advantage is that the final product can be positioned as a high-experience, tech-driven dosage form, supporting differentiated pricing.
Q4: I only have a functional idea and no formulation. Can I still start?
A: Yes. Typically, the factory or a technical team will perform formulation development based on your target market, function, flavour and price band, then move into sampling and scale-up.
Q5: What is the real difference between oral film and gummies / capsules?
A: Mainly the user experience:
No swallowing; it dissolves in the mouth.
No water required; you can use it anywhere.
More precise, smaller doses are possible.
Much easier to build a “techy + lifestyle” brand story.
Q6: How many products can one ODF production line handle?
A: As long as cleaning and cross-contamination control are well managed, a continuous ODF production line can run multiple products. Usually, products are grouped by functional module and formulation compatibility for scheduling.
Q7: What should I consider if I want to export to Europe or North America?
A: You need to check:
Ingredient legality, especially for functional actives.
Whether the product is classified as food, dietary supplement or medicine.
Labelling language, nutrition facts, and health claim rules.
Whether the manufacturing site meets the relevant GMP / hygiene standards.
Q8: I’m just a foreign-trade salesperson with no factory background. Can I really do ODF projects?
A: Yes. ODF projects are very suitable for “project-manager-type” foreign-trade professionals.
As long as you:
Can clearly explain the process using this flowchart.
Coordinate buyer needs, factory implementation and timelines.
Are willing to learn more about formulation, packaging and compliance,
you will be seen not just as “the person who sends prices”, but as the person who can actually make the project happen.